The infrared (IR) absorption process involves the absorption of light with low photon energy (from approx. 800 nm to approx. 1000 m) which promotes the excitation of molecular vibrations. Of the entire IR photon energy range, the photons which excite fundamental vibrations of covalent bonds have energies in the range 2.5-25 m Norman et al. (2018)Centrone (2015). The absorbed photon energies (i.e., IR absorption peak positions) correspond to the molecular vibrational frequencies, while the IR intensities are related to the IR linear absorption cross-section Norman et al. (2018)
Here, is the molecular dipole moment of the electronic ground state, is the normal coordinate corresponding to the vibrational mode , is the angular frequency of the absorbed electromagnetic radiation, is the angular frequency difference between the excited vibrational state and the ground state, and is the half-width broadening associated with the inverse lifetime of . As can be seen from the equation, the IR absorption cross-section depends on the derivative of the dipole moment with respect to the normal coordinate . This means that the only modes which have non-zero IR absorption cross-sections are those associated with a change in the dipole moment Siebert & Hildebrandt (2007).
These molecular vibrations constitute fingerprints of different functional groups present in the system and thus are important for chemical characterization and molecular identification.
The first step in an IR spectrum calculation is to determine the normal modes and frequencies. These are obtained from the eigenstates and eigenvalues of the Hessian matrix which collects all the second order derivatives of the energy with respect to the nuclear coordinates. For the ground state, the Hessian can be determined analytically, or numerically based on the analytical gradient. The dipole moment gradient, required to compute IR absorption cross-sections, can also be determined analytically.
import numpy as np
import py3Dmol as p3d
import veloxchem as vlx
from veloxchem.veloxchemlib import bohr_in_angstroms# Define the molecule and basis set
molecule_string = """3
O 0.000 0.000 0.000
H 0.000 0.000 0.950
H 0.896 0.000 -0.317"""
molecule = vlx.Molecule.read_xyz_string(molecule_string)
basis = vlx.MolecularBasis.read(molecule, "sto-3g", ostream=None)scf_drv = vlx.ScfRestrictedDriver()
scf_results = scf_drv.compute(molecule, basis)Now we can run vibrational analysis
vibanalysis_drv = vlx.VibrationalAnalysis(scf_drv)
vib_results = vibanalysis_drv.compute(molecule, basis)vibanalysis_drv.print_hessian(molecule) Analytical Hessian (Hartree/Bohr**2)
--------------------------------------
Coord. 1 O(x) 1 O(y) 1 O(z) 2 H(x) 2 H(y) 2 H(z)
1 O(x) 0.77341754 0.00000000 -0.22069513 -0.05793219 0.00000000 0.05915305
1 O(y) 0.00000000 -0.08462393 -0.00000000 0.00000000 0.04250781 0.00000000
1 O(z) -0.22069513 -0.00000000 0.93182618 -0.07097759 0.00000000 -0.79549474
2 H(x) -0.05793219 0.00000000 -0.07097759 0.05352688 -0.00000000 -0.01075530
2 H(y) 0.00000000 0.04250781 0.00000000 -0.00000000 -0.02940298 -0.00000000
2 H(z) 0.05915305 0.00000000 -0.79549474 -0.01075530 -0.00000000 0.80288015
3 H(x) -0.71548535 -0.00000000 0.29167272 0.00440531 -0.00000000 -0.04839775
3 H(y) -0.00000000 0.04211612 0.00000000 0.00000000 -0.01310483 -0.00000000
3 H(z) 0.16154207 0.00000000 -0.13633144 0.08173289 0.00000000 -0.00738541
Coord. 3 H(x) 3 H(y) 3 H(z)
1 O(x) -0.71548535 -0.00000000 0.16154207
1 O(y) -0.00000000 0.04211612 0.00000000
1 O(z) 0.29167272 0.00000000 -0.13633144
2 H(x) 0.00440531 0.00000000 0.08173289
2 H(y) -0.00000000 -0.01310483 0.00000000
2 H(z) -0.04839775 -0.00000000 -0.00738541
3 H(x) 0.71108004 0.00000000 -0.24327497
3 H(y) 0.00000000 -0.02901129 -0.00000000
3 H(z) -0.24327497 -0.00000000 0.14371685
The normal modes and frequencies are then determined by diagonalizing the Hessian. First, the translation and rotational modes must be projected out, leaving (or for a linear molecule) vibrational modes for a system with atoms. This is done under the hood with the help of geomeTRIC.
The IR intensities are determined as discussed in more detail here.
def add_broadening(
list_ex_energy,
list_osci_strength,
line_profile="Lorentzian",
line_param=10,
step=10,
):
x_min = np.amin(list_ex_energy) - 50
x_max = np.amax(list_ex_energy) + 50
x = np.arange(x_min, x_max, step)
y = np.zeros((len(x)))
# go through the frames and calculate the spectrum for each frame
for xp in range(len(x)):
for e, f in zip(list_ex_energy, list_osci_strength):
if line_profile == "Gaussian":
y[xp] += f * np.exp(-(((e - x[xp]) / line_param) ** 2))
elif line_profile == "Lorentzian":
y[xp] += (
0.5
* line_param
* f
/ (np.pi * ((x[xp] - e) ** 2 + 0.25 * line_param**2))
)
return x, y# plot the IR spectrum
from matplotlib import pyplot as plt
plt.figure(figsize=(7, 4))
x1, y1 = vibanalysis_drv.vib_frequencies, vibanalysis_drv.ir_intensities
x1i, y1i = add_broadening(x1, y1, line_profile="Gaussian", line_param=20, step=10)
plt.plot(x1i, y1i)
plt.xlabel("wavenumber (cm**-1)")
plt.ylabel("IR intensity (km/mol)")
plt.title("Calculated IR sepctrum of the water molecule")
plt.tight_layout()
plt.show()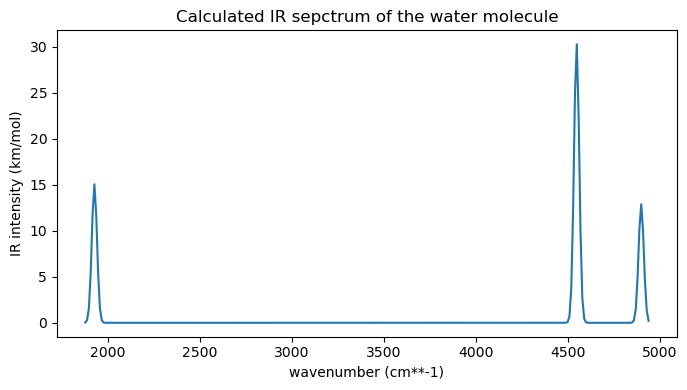
Let’s now visualize the three vibrational normal modes. The two high frequency modes are OH stretching modes (one symmetryc and one asymmetric), while the lower energy mode is a bond angle bending.
# To animate the normal mode we will need both the geometry and the displacements
def get_normal_mode(molecule, normal_mode):
elements = molecule.get_labels()
# Transform from au to A
coords = molecule.get_coordinates_in_bohr() * bohr_in_angstroms()
natm = molecule.number_of_atoms()
vib_xyz = "%d\n\n" % natm
nm = normal_mode.reshape(natm, 3)
for i in range(natm):
# add coordinates:
vib_xyz += elements[i] + " %15.7f %15.7f %15.7f " % (
coords[i, 0],
coords[i, 1],
coords[i, 2],
)
# add displacements:
vib_xyz += "%15.7f %15.7f %15.7f\n" % (nm[i, 0], nm[i, 1], nm[i, 2])
return vib_xyzvib_1 = get_normal_mode(molecule, vibanalysis_drv.normal_modes[0])
vib_2 = get_normal_mode(molecule, vibanalysis_drv.normal_modes[1])
vib_3 = get_normal_mode(molecule, vibanalysis_drv.normal_modes[2])print("This is the bending mode at 1927.09 cm-1.")
view = p3d.view(width=300, height=300)
view.addModel(vib_1, "xyz", {"vibrate": {"frames": 10, "amplitude": 0.75}})
view.setViewStyle({"style": "outline", "width": 0.05})
view.setStyle({"stick": {}, "sphere": {"scale": 0.25}})
view.animate({"loop": "backAndForth"})
view.rotate(-90, "x")
view.zoomTo()
view.show()This is the bending mode at 1927.09 cm-1.
print("This is the symmetric stretching mode at 4545.78 cm-1.")
view = p3d.view(width=300, height=300)
view.addModel(vib_2, "xyz", {"vibrate": {"frames": 10, "amplitude": 0.75}})
view.setViewStyle({"style": "outline", "width": 0.05})
view.setStyle({"stick": {}, "sphere": {"scale": 0.25}})
view.animate({"loop": "backAndForth"})
view.rotate(-90, "x")
view.zoomTo()
view.show()This is the symmetric stretching mode at 4545.78 cm-1.
print("This is the asymmetric stretching mode at 4896.44 cm-1.")
view = p3d.view(width=300, height=300)
view.addModel(vib_3, "xyz", {"vibrate": {"frames": 10, "amplitude": 0.75}})
view.setViewStyle({"style": "outline", "width": 0.05})
view.setStyle({"stick": {}, "sphere": {"scale": 0.25}})
view.animate({"loop": "backAndForth"})
view.rotate(-90, "x")
view.zoomTo()
view.show()This is the asymmetric stretching mode at 4896.44 cm-1.
- Norman, P., Ruud, K., & Saue, T. (2018). Principles and practices of molecular properties. John Wiley & Sons, Ltd.
- Centrone, A. (2015). Infrared imaging and spectroscopy beyond the diffraction limit. Annu. Rev. Anal. Chem., 8, 101–126. 10.1146/annurev-anchem-071114-040435
- Siebert, F., & Hildebrandt, P. (2007). Theory of Infrared Absorption and Raman Spectroscopy. In Vibrational Spectroscopy in Life Science (pp. 11–61). John Wiley. 10.1002/9783527621347.ch2